Some people consider mosquitoes “the most dangerous animals in the world.” It’s hard to imagine. It is true that mosquitoes do not enjoy a good reputation. They can ruin our afternoon in the garden or a barbecue. Or wake up in the middle of the night. They are annoying. This is beyond doubt, but what makes them so dangerous?
Its danger lies in the diseases it can transmit. Mosquitoes are potential transmitters of diseases such as malaria, dengue, yellow fever and Zika, which affect hundreds of millions of people worldwide each year. It is estimated that 390 million people are infected with the dengue virus each year. In 2018, it was estimated that there were 228 million cases of malaria. The World Health Organization estimates that more than half of the world’s population is at risk of contracting mosquito-borne diseases. This ability to transmit viruses and other pathogens through their bites is what makes them dangerous, making their study, surveillance and control necessary.
The mosquitoes that we study and monitor in Mosquito Alert with the collaboration of the citizens stand out for being able to transmit these diseases:
Chikungunya (arbovirus)
Yellow fever (arbovirus)
West Nile fever (arbovirus)
Dirofilariasis (parasitic nematode)
With the exception of malaria, most mosquito-borne diseases that affect people are of viral origin. Viruses that are transmitted through mosquitoes and other arthropods are called “arboviruses.” The term encompasses different families of viruses that are characterized by being transmitted between vertebrate animals (including humans) by an arthropod. Mosquitoes transmit specific viruses, but other arboviruses are transmitted by bedbugs, ticks, sandflies, and other arthropods.
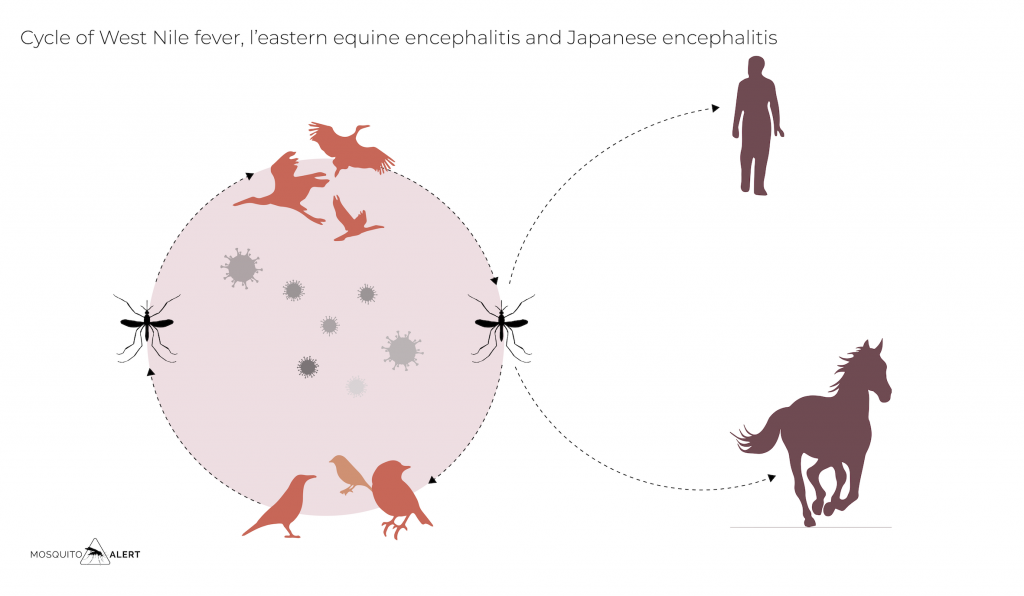
They are all hematophagous animals, that is, they feed on the blood of other animals, usually vertebrates, such as mammals, birds, reptiles or amphibians. This figure represents a typical cycle of an arbovirus transmitted by a mosquito.
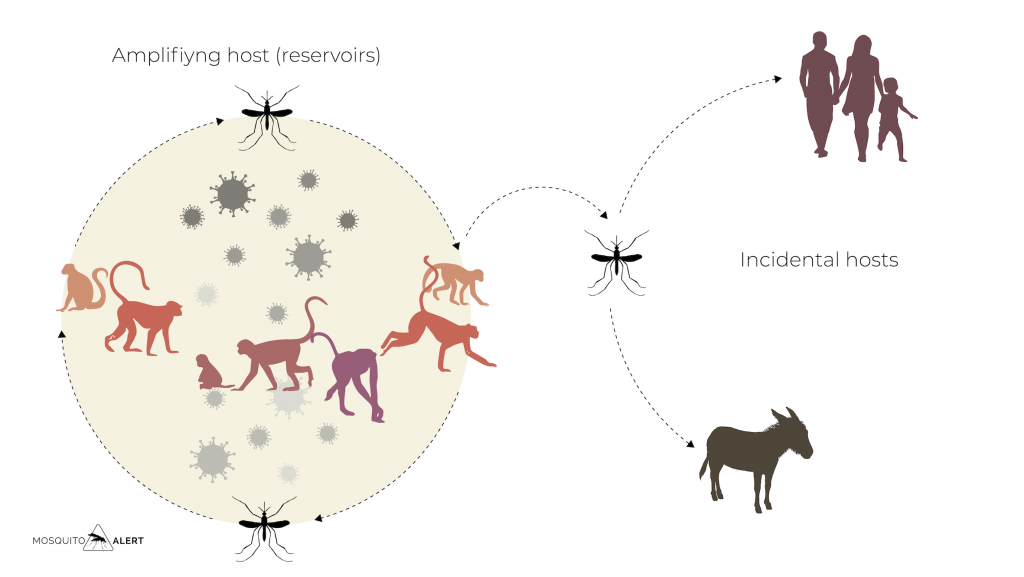
For an arbovirus to maintain its cycle, the following conditions in nature must be met: (1) a susceptible vertebrate host, possibly a mammal or a bird, must be present; (2) during the infection of the host, a viremia phase must be reached (ie, the virus is present in the bloodstream in sufficient quantity and time for the mosquito to become infected when it feeds on its blood); (3) the virus must be able to replicate and not kill the mosquito that has ingested the blood; (4) After a few days of incubation, the mosquito may infect another animal or person by re-feeding. At that time, it will inject saliva from its infected glands and the virus will enter the new host’s bloodstream to begin the cycle.
Vertebrates with high levels of viremia sufficient for mosquitoes to become infected by biting them are known as amplifying hosts, in the figure above they are represented as primates. Those who do not become infected or do not develop enough viremia to infect other mosquitoes are known as incidental or “dead-end” hosts, as the virus cannot be transmitted to anyone else from them, this host being a dead end for the virus.
Humans are usually incidental hosts in most arboviruses, whose amplifying hosts are other mammals or birds. However, in the case of dengue, Zika and chukingunya, people can act as the main amplifying host, jumping from one person to another by biting a mosquito.
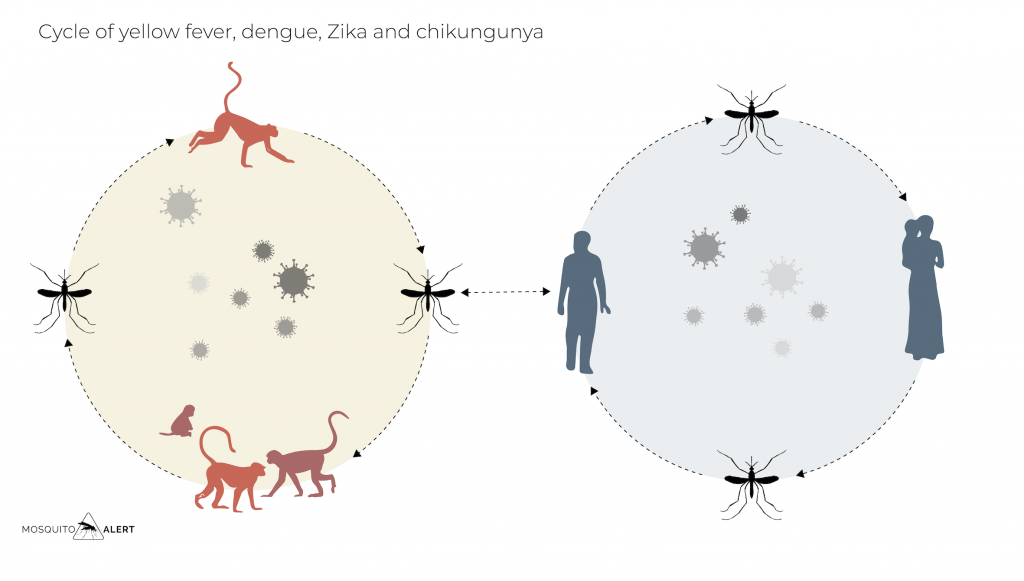
Mosquitoes are thus the vehicles that allow viruses and other pathogens to circulate between people, between vertebrate animals, and between animals and people. Fortunately, most 3,500 species of mosquitoes are not able to transmit viruses, and among the few species that can do so, their effectiveness varies greatly between species. Species such as the yellow fever mosquito (Aedes aegypti) and the tiger mosquito (Aedes albopictus) are very effective in transmitting some of the viruses that most affect people. Other mosquitoes are not as effective at transmitting viruses. Most are not able to become infected and transmit these viruses.
Some species have the ability to transmit different viruses, others only effectively transmit one or two viruses that affect humans. The yellow fever mosquito (Aedes aegypti) is able to transmit dengue virus, yellow fever virus, chikungunya virus and Zika virus, some studies suggesting that it can also transmit West Nile virus and Venezuelan equine encephalitis virus. The tiger mosquito (Aedes albopictus) in the laboratory is able to become infected with 22 different viruses, several of which have been detected in mosquitoes analyzed in the field. However, others do not have such a large capacity, or their effectiveness with the various viruses is much lower, and therefore of less medical concern, as would be the case with Aedes japonicus and Aedes koreicus.
The complexity of the arbovirus cycle means that those that affect human populations show different patterns. From sporadic infections with one or a few cases, to generating large epidemics.
The climate determines the distribution of arboviruses
As these viruses require mosquitoes to circulate between animals and humans, it is normal for climatic and ecological factors to influence the presence and transmission of diseases. Temperature, rain and humidity are very important factors in the life of mosquitoes and, therefore, of the transmission of the virus. In the tropics, mosquitoes are active mainly during the wet season, when rains provide places for the development of their larvae. In Europe and other temperate regions, mosquito activity is seasonal, with its maximum activity concentrated in summer.
All predictions suggest that climate change will affect worldwide transmission cycles. Rising temperatures and changes in rainfall will allow disease-transmitting mosquitoes to spread to new areas. Human activities also have an impact on the distribution of mosquitoes, with the destruction of some habitats, the increase in urbanism and their water storage strategies that can benefit mosquitoes adapted to human populations.
The globalization of arboviruses
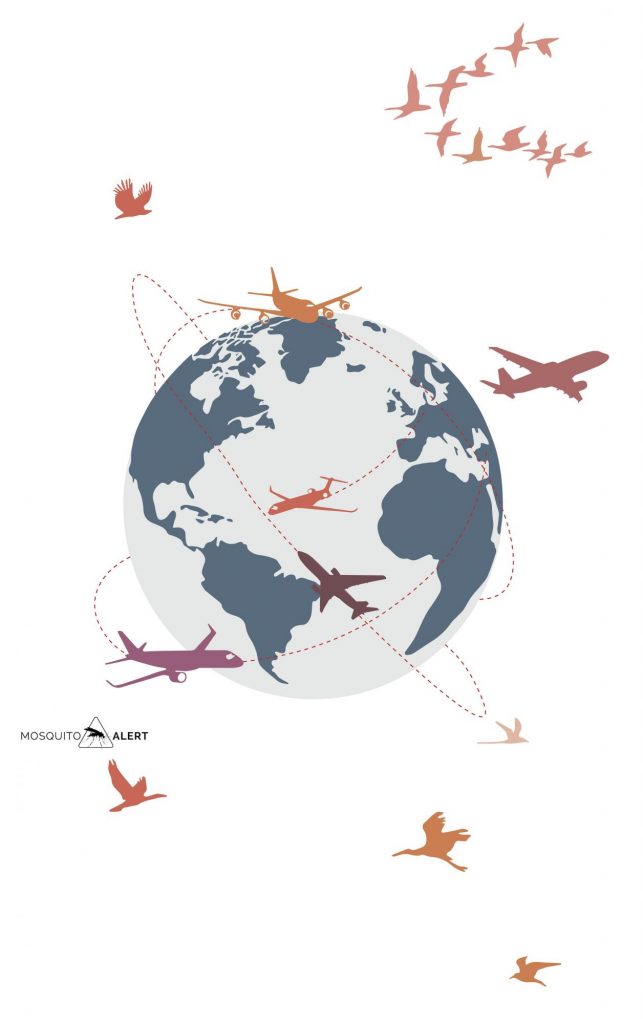
Globalization and environmental change have led to an increase in the distribution and impact of arboviruses in the last century. New invasive species, such as the tiger mosquito (Aedes albopictus) have spread around the world in just half a century. It has been expanding in Europe since 1979, as have other more recently arrived Asian species of Aedes (Aedes japonicus and Aedes koreicus). Its global expansion, along with that of Aedes aegypti, has meant that in recent decades the population susceptible to dengue infection has increased considerably in the world. In half a century, dengue cases have multiplied by 30. In the 1960s there were 10 countries with endemic dengue, in 2017 there were already 124 countries.
The expansion of these invasive species has created a new epidemiological scenario. In regions where the virus is not endemic, such as Europe, isolated cases of dengue have been reported in several Mediterranean countries (Croatia, France, Spain). Local transmission is possible thanks to the presence of the tiger mosquito, which is able to become infected with the virus when it bites a traveler who has become infected from other regions. That mosquito can later infect another person. The same has happened with the chikungunya or Zika virus. In Madeira, Aedes aegypti starred in an outbreak of dengue that affected more than 2,000 people, being the origin of 78 imported cases that were later detected in 13 different countries. Despite being a virus-free region, the global expansion of dengue is making an impact in Europe.
The speed of current travel, and the increase in destinations to tropical or subtropical areas, allow diseases with short incubation periods to be exported to virus-free regions such as Europe. One study found that 40% of passengers returning with dengue in Europe had virus levels in their bloodstream high enough for a mosquito to become infected.
Other diseases such as West Nile fever have established themselves in Europe. Birds are its main amplifying hosts, and the disease reached the European continent with migratory birds returning after migrating to Africa. Since the end of the twentieth century, the virus has circulated, both among migratory birds and in local populations, occasionally infecting people and horses. The mosquitoes that transmit this virus are not invasive mosquitoes, but native mosquitoes of the genus Culex, including the common house mosquito, Culex pipiens.
Urbanization and domestication of mosquitoes
The global resurgence and expansion of various arboviruses is not only due to the increase in transportation systems in recent decades, but also to the urbanization experienced by the planet. That has allowed people to concentrate on certain areas. High densities favor the transmission of diseases. Changes in density and mobility in the last century have altered the virus-mosquito-human interaction.
Population growth and urbanization have been accompanied by increased agricultural, livestock and deforestation capacity. Humans have been occupying new forest areas, altering their ecosystems and coming into contact with mosquitoes and viruses much more often. Mosquitoes are increasingly exposed to more humane environments, with buildings, water tanks, humans and livestock. Their fast reproduction cycles allow them to adapt quickly to these new circumstances. This process is often called “domestication.” Mosquitoes are self-domesticated to live in our environments.

Reviewing the history of the dengue virus, yellow fever, Zika and chikungunya, it is observed that all four exhibit similar ecological, evolutionary and epidemiological patterns in their expansion around the world. They all originate in African forests, with initial cycles involving Aedes aegypti and other Aedes species, with primate hosts. The viruses, like the Aedes aegypti mosquito, have been accidentally transported across the Atlantic and Pacific to all continents. All viruses have exploited the domestic form of the Aedes aegypti mosquito, which at one time adapted to grow and feed in urban environments. Populations of the species with a preference for entering homes and feeding on humans. Consequently, all these viruses can be kept transmitted between humans without depending on the animals and forest cycles from which they arose.
The tiger mosquito (Aedes albopictus), is another species native to forests, in its case Southeast Asia, which like the mosquito Aedes aegypti has adapted to live in urban areas. International trade has allowed its spread across all continents in a few decades, playing a supporting role in the transmission of these viruses in semi-urban and rural environments.
References and complementary readings:
Bhatt S, Gething PW, Brandy O, […] Hay S. 2013. The global distribution and burden dengue. Nature 496: 504-507
Brugueras S, Fernández-Martínez B, Martínez-de la Puente, […] Gómez-Barroso D. 2020. Environmental drivers, climate change and emergent diseases transmitted by mosquitoes and their vectors in Southern Europe: a systematic review. Environmental Research 191: 110038
Ebi KL, Nealon J. 2016. Dengue in a changing climate. Environmental Research 151: 115-123
Gould EA, Higgs S. 2009. Impact of climate change and other factors on emerging arbovirus diseases. Transactions of The Royal Society of Tropical Medicine and Hygiene 103: 109-121
Gould E, Pettersson J, Higgs S, Charrel R, de Lamballerie X. 2017. Emerging arbovirus: why today? One Health 4: 1-13
Liang G, Gao X, Gould EA. 2015. Factors responsible for the emergence of arboviruses; strategies, challenges and limitations for their control. Emerging Microbes & Infections 4: /10.1038/emi.2015.18
Massad E, Amaku M, Coutinho FAB, Struchiner CJ, Burattini MN, Khan K, Liu-Helmersson J, Rocklöv J, Kraemer MUG, Wilder-Smith A. 2018. Estimating the probability of dengue virus introduction and secondary autochthonous cases in Europe. Scientific Reports 8: 4629
Murray NEA, Quam MB, Wilder-Smith A. 2013. Epidemiology of dengue: past, present and future prospects. Clinical Epidemiology 5: 299-309
Neumayr A, Muñoz J, Shunk M, […] Franco L, TropNet. 2017. Sentinel sruveillance of imported dengue travellers to Europe 2012 to 2014: TropNet data from the Dengue Tools Research Initiative. EuroSurveillance 22
Sejvar JJ. 2003. West Nile Virus: an historial overview. The Ochsner Journal 5: 6-10